Question
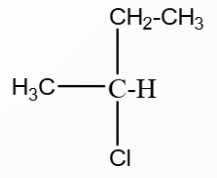
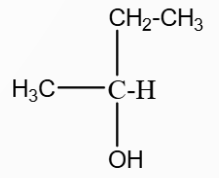
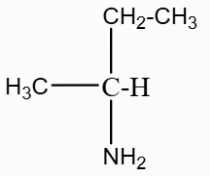
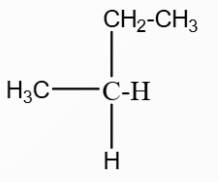
A hydrocarbon $A(C_4H_8)$ on reaction with $HCl$ gives a compound $B(C_4H_9Cl)$, which on reaction with 1 mol of $NH_3$ gives compound $C(C_4H_{10}N)$. On reacting with $NaNO_2$ and $HCl$ followed by treatment with water, compound $C$ yields an optically active compound $D$. The $D$ is