Question
One mole of an ideal gas is taken from A to B, from B to C, and then back to A. The variation of its volume with temperature for that change is as shown. Its pressure at A is $P_0$, and its volume is $V_0$. Then, the internal energy
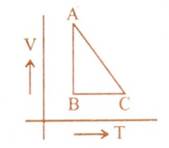
One mole of an ideal gas is taken from A to B, from B to C, and then back to A. The variation of its volume with temperature for that change is as shown. Its pressure at A is $P_0$, and its volume is $V_0$. Then, the internal energy
