Question
Three energy levels of a hydrogen atom and the corresponding wavelengths of the emitted radiation due to different electron transitions are shown. Then:
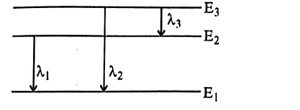
Three energy levels of a hydrogen atom and the corresponding wavelengths of the emitted radiation due to different electron transitions are shown. Then:
