Question
For a reaction consider the plot of $\ell $n k versus 1/T given in the figure. If the rate constant of this reaction at 400 K is 10–5 s–1, then the rate constant at 500 K is –
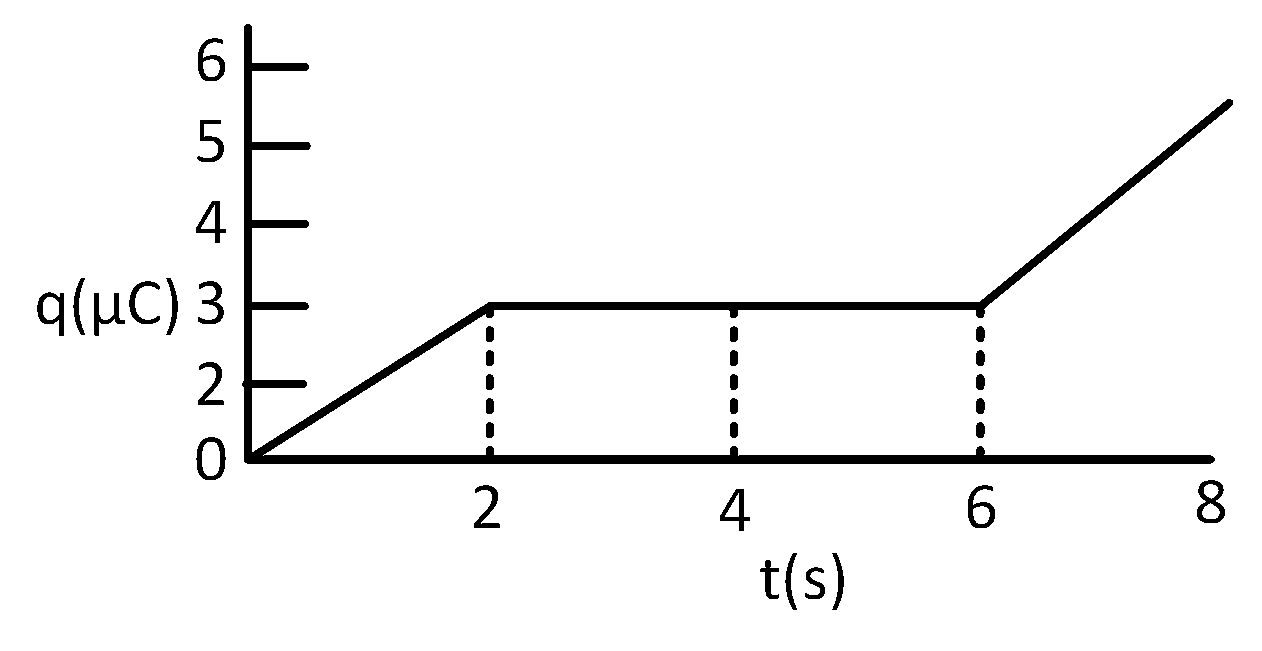
For a reaction consider the plot of $\ell $n k versus 1/T given in the figure. If the rate constant of this reaction at 400 K is 10–5 s–1, then the rate constant at 500 K is –
